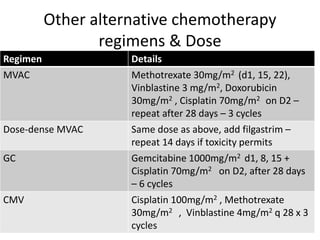
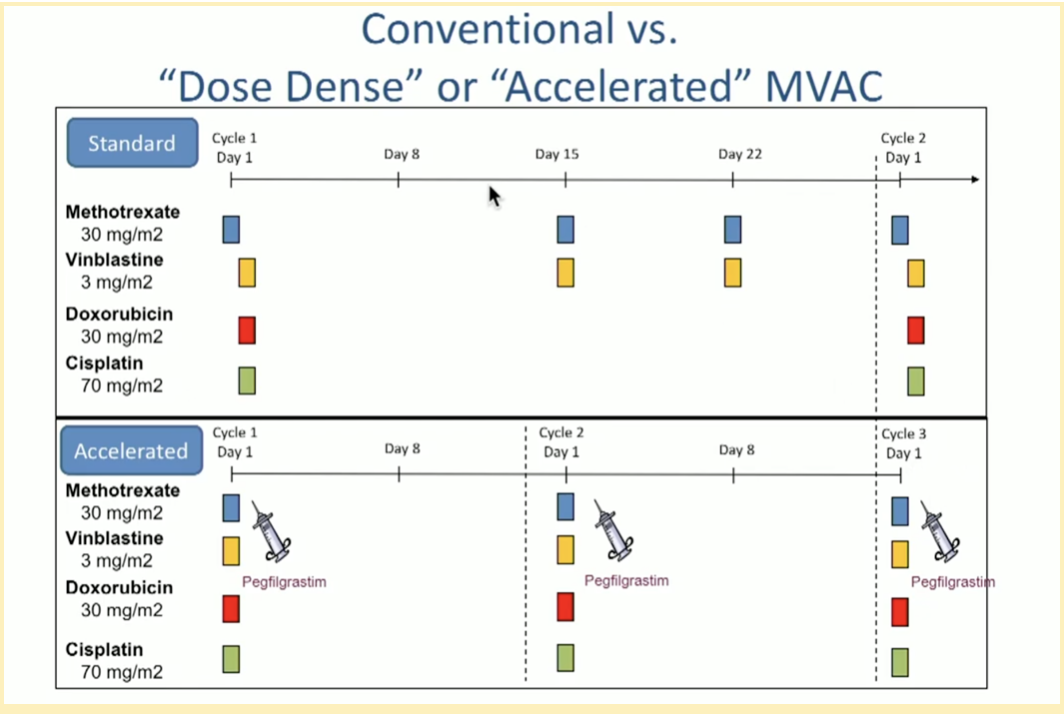
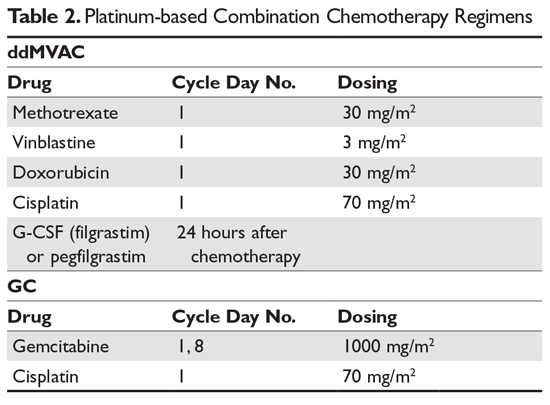
Prospective, open-label, randomized, phase III study of two dose-dense regimens MVAC versus gemcitabine/cisplatin in patients with inoperable, metastatic or relapsed urothelial cancer: a Hellenic Cooperative Oncology Group study (HE 16/03). | Semantic
Prospective, open-label, randomized, phase III study of two dose-dense regimens MVAC versus gemcitabine/cisplatin in patients wi

ESMO 2021: Avelumab as the Basis of Neoadjuvant Chemotherapy Regimen in Platinum Eligible and Ineligible Patients with Non-Metastatic Muscle Invasive Bladder Cancer (NM-MIBC)

Cancers | Free Full-Text | Comparison of Oncologic Outcomes of Dose-Dense Methotrexate, Vinblastine, Doxorubicin, and Cisplatin (ddMVAC) with Gemcitabine and Cisplatin (GC) as Neoadjuvant Chemotherapy for Muscle-Invasive Bladder Cancer: Systematic ...
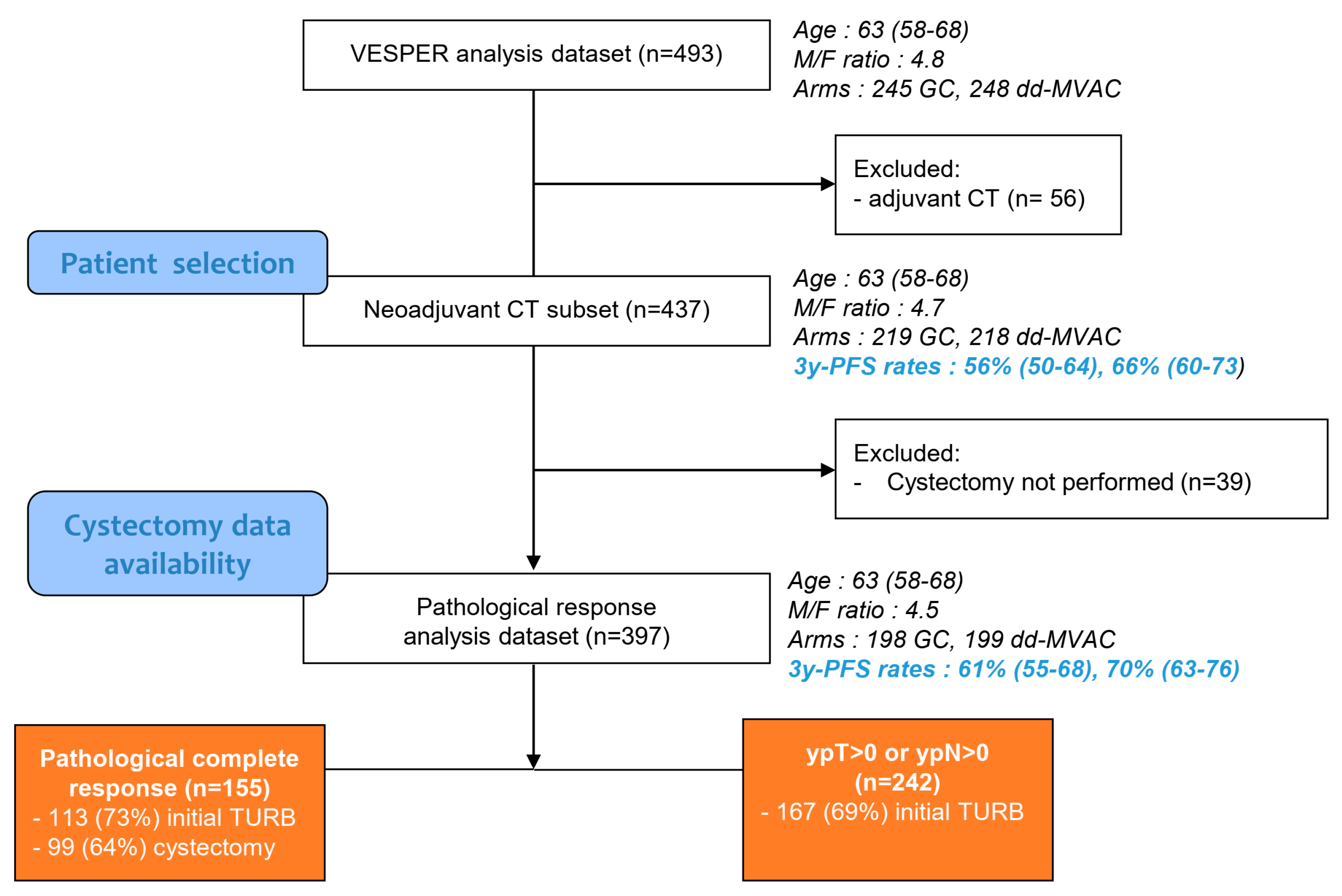
Cancers | Free Full-Text | Refining the Characterization and Outcome of Pathological Complete Responders after Neoadjuvant Chemotherapy for Muscle-Invasive Bladder Cancer: Lessons from the Randomized Phase III VESPER (GETUG-AFU V05) Trial

Randomized phase III trial of dose-dense MVAC or GC as perioperative chemotherapy for muscle-invasive urothelial bladder cancer (MIUBC): Preliminary results of the GETUG/AFU V05 VESPER trial on toxicity and pathological responses -

Prospective, open-label, randomized, phase III study of two dose-dense regimens MVAC versus gemcitabine/cisplatin in patients with inoperable, metastatic or relapsed urothelial cancer: a Hellenic Cooperative Oncology Group study (HE 16/03). | Semantic
Neoadjuvant chemotherapy with dose dense MVAC is associated with improved survival after radical cystectomy compared to other cy

Prospective, open-label, randomized, phase III study of two dose-dense regimens MVAC versus gemcitabine/cisplatin in patients with inoperable, metastatic or relapsed urothelial cancer: a Hellenic Cooperative Oncology Group study (HE 16/03). | Semantic
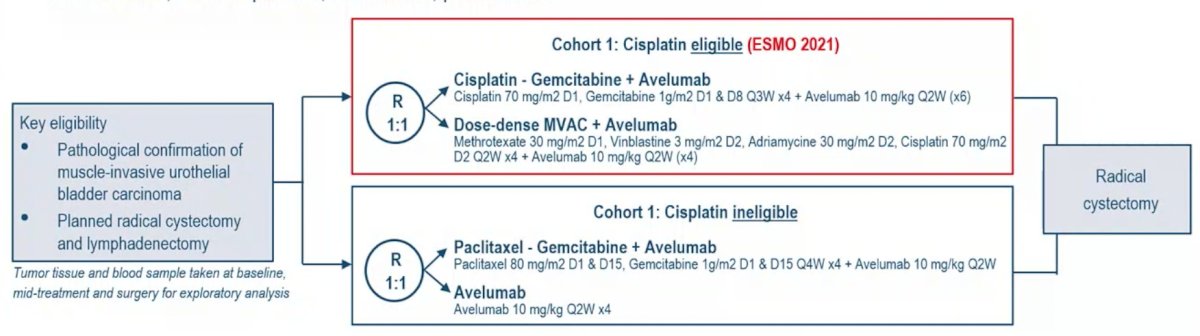
ESMO 2021: Avelumab as the Basis of Neoadjuvant Chemotherapy Regimen in Platinum Eligible and Ineligible Patients with Non-Metastatic Muscle Invasive Bladder Cancer: AURA Trial